C.S. Young1, S. Terada2, J.P. Vacanti2, M. Honda3, J.D. Bartlett1*, and P.C. Yelick1* 1Department of Cytokine Biology and Harvard-Forsyth Department of Oral Biology, The Forsyth Institute, Boston, MA 02115, USA; 2Department of Surgery, Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA; and 3Department of Oral and Maxillofacial Surgery, Nagoya University School of Medicine, Nagoya, Japan; *corresponding authors, pyelick@forsyth.org, jbartlett@forsyth.org | J Dent Res 81(10):695-700, 2002 (http://jdr.iadrjournals.org/cgi/content/abstract/81/10/695)
Wh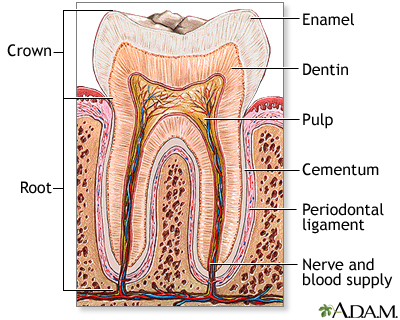 y Study Tooth Tissue Engineering? (other than the fact that teeth are simply awesome!)
y Study Tooth Tissue Engineering? (other than the fact that teeth are simply awesome!)
Trauma, dental caries, and a variety of genetic disorder such as amelogenesis imperfecta can result in a tooth loss, for which there is no regenerative biological substitute currently. This particular area of research has been flourishing since the Forsyth Institute (Boston, MA) presented a possibility of generating bioengineered dental tissues after they found that bioengineered dental tissues were constructed using mixed population of cultured post-natal tooth bud cells (British Dental Journal (2006); 201, 73. doi: 10.1038/sj.bdj.4813885). In order to investigate a tissue engineering approach to construct a biologically suitable substitute for a tooth, this paper I chose (coincidentally it’s from the Forsyth Institute) examines a tissue engineering approach that was used to bioengineer small intestines successfully (Choi and Vacanti, 1997).
What does this study aim to discover in order to demonstrate a regenerative tooth crown?
The signaling between dental mesenchyme that differentiates into pulp and dentin tissues and epithelial tissue of the enamel that produces the dental enamel plays a pivotal role in the overall development of teeth: allowing each tooth to possess different characteristics such as its specific size, shape and function within different positions in the jaw. Thus, this study aims to find the presence of epithelial and mesenchymal dental stem cells in porcine third molar tissues that allow for a successful construction of tooth crowns from dissociated tooth tissues.
*Summary*
Biodegradable polymer scaffolds (a human tooth-shaped polymer scaffolds made with PGA/PLLA and PLGA) were used as substrates to seed dissociated cells from tooth tissues. Third molar tooth buds were obtained from 6 months old pig jaws; pulp organ tissues and enamel tissues were minced after. This cell/polymer constructs were implanted into a suitable host (rat) to allow for the growth of higher-ordered structures. Histological, immunohistochemical, and Laser-capture Microdissection and Reverse-transcription/Polymerase Chain-reaction (LCM, RT-PCR) analyses were on samples from 20, 25, and 30-weeks of implants.
Overall, molecular evidences found from the noted analytical methods indicated a successful bioengineering of complex tooth crowns that closely resemble naturally developing teeth. After 20-weeks of implanting, mineralized dentin, pre-dentin, and pulp tissue resembling vascularized mesenchyme were present. Amelogenin, one of the most prominent extracellular matrix proteins in developing enamel was immunodetected in the enamel matrix of 25-week implant as well as the presence of ameloblast-like cells adjacent to the enamel. 30-week implant tissues showed a thick layer of enamel surrounding a layer of dentin. Amelogenin was detected at the secretory ends of ameloblast-like cells, confirming the occurrence of amelogenesis in the developing enamel. LCM and RT-PCR using nested primers for odontoblast-specific DSPP mRNA confirmed the identity of putative odontoblast-like cells present in bioengineered tooth tissues. Given these morphological, histological, and immunochemical evidences from 20-, 25-, and 30-week implant tissues, this study overall demonstrated a successful construction of bioengineered teeth containing the similar organization and molecular markers of natural teeth such as dentin, pulp, and amelogenin proteins.
Why Did I Choose this Paper?
The approach (seeding dissociated tooth tissues onto biodegradable scaffolds) this paper took signifies a possibility of generating teeth of preprogrammed size and shape, giving engineers more control over the overall function and morphology of bioengineered tooth. The bioengineered tissues not only provided morphologically evidences of natural teeth, LCM and RT-PCR further confirmed the on-going process of amelogenesis. Since my research interest lies within dental science, specifically in understanding amelogensis by closely mimicking the enamel formation in vitro, I chose this paper to study possible approaches engineers can take in regenerating teeth that closely resemble the natural teeth. If we can understand how biological system is able to design organized structures (ie. enamel) in tissues at the molecular level, not only people with genetically impaired teeth could recover their full dentition but we may even be able to use artificial human enamel to build a dental restoration. For instance, if the molecular mechanism of amelogensis was known, engineers can further manipulate the developmental process of bioengineered teeth to produce fully-developed enamel.
*Here are some interesting papers you can read at your leisure:
- Micelle Structure of Amelogenin in Porcine Secretory Enamel: J Dent Res 86(8):758-763, 2007 (http://jdr.iadrjournals.org/cgi/content/abstract/86/8/758)
- Bioengineered Teeth from Cultured Rat Tooth Bud Cells: J Dent Res 83(7): 523-528, 2004 (http://jdr.iadrjournals.org/cgi/content/full/83/7/523)
- New observations of the hierarchical structure of human enamel, from nanoscale to microscale: Journal of Tissue Engineering and Regenerative Medicine V1 I3: 185-191 (http://www3.interscience.wiley.com/cgi-bin/abstract/114219401/ABSTRACT)



